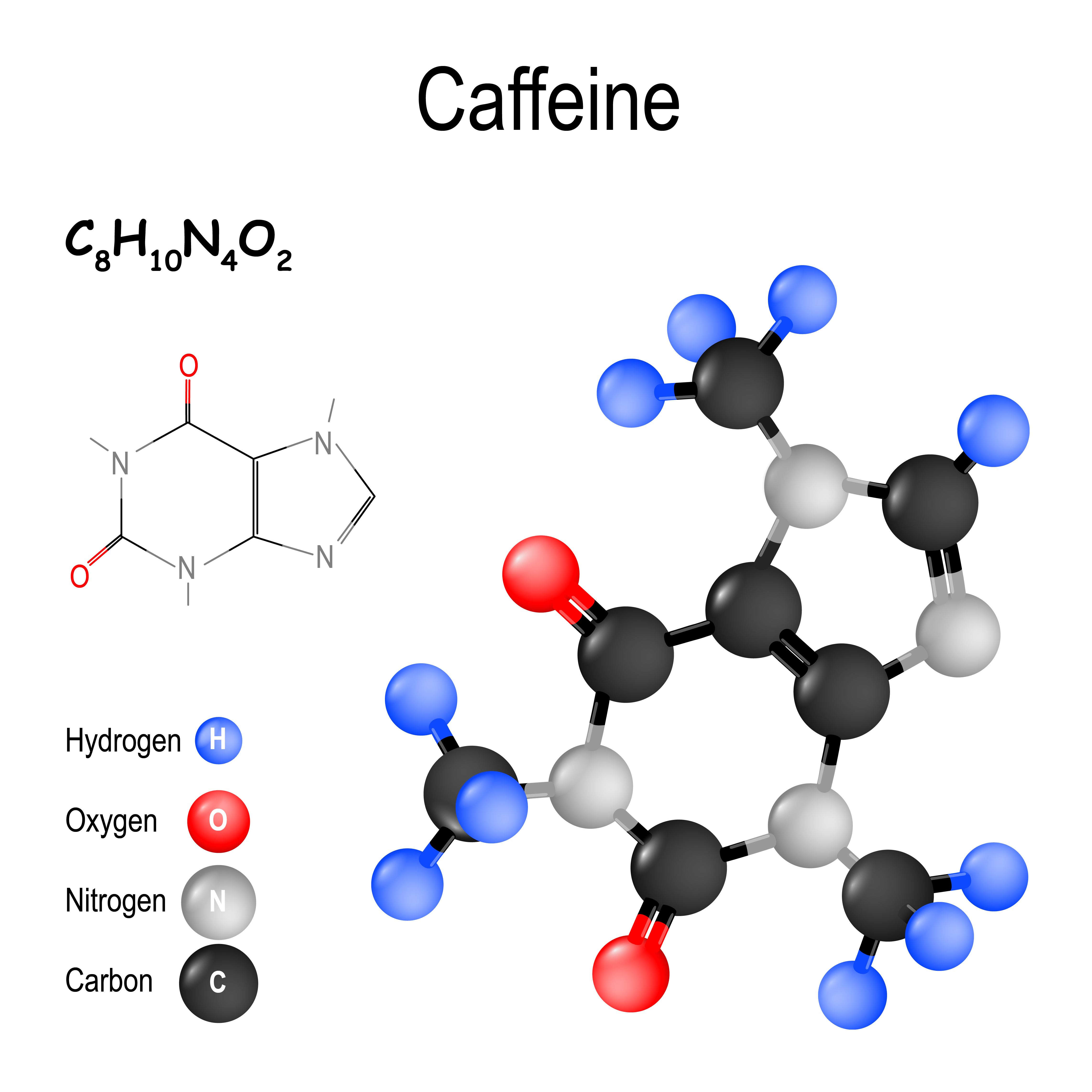
Examples of isomers: The table below gives the number of total electrons for each element in blue and the number of valence electrons in red. Lewis structures can represent Isomers which are molecules with the same molecular formulas but have different arrangements of atoms.show valence electrons that are not used for bonding as dots called unshared pairs or lone pairs. 
display the bond order (single, double.they show the connectivity of between atoms with dots or dashes representing bonding electrons.Lewis structures represent chemical substances by showing bonds formed by valence electrons.Name: 2 Lewis Structures ptable/?lang=en#Properties Lewis diagram of a caffeine molecule Notes: Lewis Dot Structures How many valence electrons are found in group 6 elements? Click on the link below to answer the following questions. Lithium 3 1 s 2 2 s 1 Beryllium 4 1 s 2 2 s 2 Oxygen 8 1 s 2 2 s 22 p 4 Fluorine 9 1 s 2 2 s 22 p 5 Neon 10 1 s 2 2 s 22 p 6 The Lewis dot structure for lithium is The Lewis dot structure for fluorine is 1. This involves research into adenosine receptors on certain immune cells, to see what would be a good target for drugs to act on.Name: 2 Lewis Structures ptable/?lang=en#Properties Lewis diagram of a caffeine molecule Notes: Lewis Dot Structures Besides that, it is still mainly happening in and around our lab, together with LUMC. A hospital wanted to investigate a disease in which adenosine seems to play a role. Is there an immediate rush of researchers using the molecules? "Six months ago, a request came in from the US. in November, his results having been published earlier. When the caffeine-like molecule is attached to the adenosine receptor, another molecule that emits light, for example, clicks onto it."īeerkens received his Ph.D. "We succeeded by using click chemistry." Last year, the Nobel Prize in chemistry went to the development of this type of chemistry. This was a tricky point in Beerkens' project. "Then researchers can see if and where which type of receptor is present." That observation also had to be doable in living cell material, that is, without high doses of radioactive radiation. The molecules not only had to bind tightly and selectively to the receptor, but also place a molecular label on it. For the fourth, my predecessor has already done it." Click chemistry turned out to be the solution, like Lego For three of them I was able to design suitable molecules. "There are four different adenosine receptors. He succeeded in making molecules that not only bind tightly to the receptor, but also only to this type of receptor and not to anything else. "Caffeine also binds to the receptor, just not very strongly."īeerkens experimented with additional atomic groups substituted onto the caffeine molecule. Caffeine became the basis of the substances he developed. To investigate this, molecules are needed that, like adenosine, bind to adenosine receptors. Some tumors abuse this mechanism by actively producing adenosine receptors."īinding firmly to it as well as labeling the receptor "Depending on the type of cell that picks up the adenosine, the result may be that the immune response is inhibited. If there is a lot of adenosine around a cell, it could mean that a cell in the area has died." Then adenosine roams around from degraded ATP, and a neighboring cell picks it up with special receptors on the cell membrane. This stores energy in all living cells and makes it available where it is needed.īeerkens says, "Adenosine is also a signaling substance for communication between cells. It is part of DNA and also of ATP: adenosine triphosphate. research, Beerkens set to work to make molecules that bind to a protein that normally binds the substance adenosine. "You then design and produce molecules that have no medicinal effect, but which help other researchers in their work on a particular condition." As a chemistry student, Bert Beerkens became fascinated by the field of research that allows chemistry to be used to study all kinds of processes in the body.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
